Publication Details:
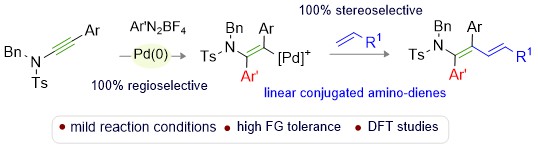
Title: Palladium-Catalyzed Regioselective Arylalkenylation of Ynamides
| Author(s) | Rajeshwer Vanjari, Shubham Dutta, Shengwen Yang, Vincent Gandon, and Akhila K. Sahoo* : Org. Lett., Org. Lett. 2022, 24, 1524–1529. |
|---|---|
| Journal Name | Organic Letters |
| journal image URL | http://chemistry.uohyd.ac.in/~aks/wp-content/uploads/OL-2022 logo.jpg |
More Details:
A cationic palladium-catalyzed arylalkenylation of ynamides is presented. The putative keteniminium arylpalladium intermediate likely dictates the regioselective carbopalladation of the ynamide to form a vinylpalladium species. The capture of this complex by the olefin yields linear conjugated β-alkenyl aminodienes (especially with trans selectivity). The transformation features a broad scope with labile functional group tolerance and makes 42 unusual molecular scaffolds with structural diversity. DFT studies provide valuable insights into the reaction mechanism.